
∆ U = molar lattice energy molar lattice energy UL denotes the equilibrium value of lattice energy. R 0 is the distance between the closest ions. Ε o = Permittivity of unoccupied free space Z+ is the charge of the cation and Z- is for anion. The Lattice Energy Formula per mole is represented by the symbolĪvogadro’s constant (6.022 1022) is denoted by the letter N A. The average of the values for Na+ and Cl– is given by n = 8. The Born-Lande equation has a radius of 2.81 x 10-10 m, which is the sum of the radii. We use the Born-Lande equation to calculate the Lattice energy of NaCl. Lattice energy is represented by the expression It should be noted that this law holds true for both cyclic and non-cyclic processes. In this case, according to Hess’s law, plus Q equals q1 plus q2 plus q3. If a chemical reaction occurs in one step or several steps, the total heat of reaction remains constant, according to Hess’ laws.įor example, in the case of a chemical reaction A B, the heat of reaction (H) equals +Q.Īlternatively, if the reaction occurs in a series of steps, as follows: The Born-Haber cycle is based on Hess’ law of constant heat of summation, which can be found in the textbooks. The magnitude of charge associated with the constituent ions and the distance between the constituent ions are the two primary factors that influence the lattice energy of an ionic compound, and they are as follows: The Born-Haber Cycle 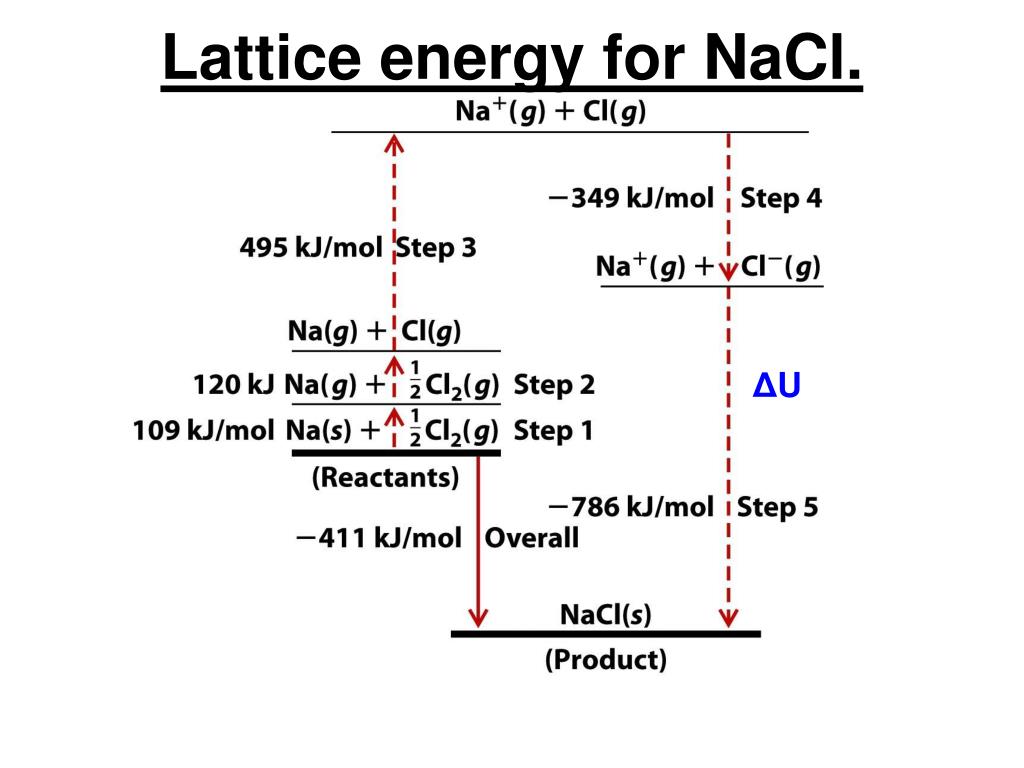
Factors that influence the amount of lattice energy available 
The change in volume is represented by the symbol Vm (per mole).Īs a result, when calculating the lattice energies of ionic solids, the outer pressure must also be taken into account. The molar lattice enthalpy is denoted by the symbol G H. The molar lattice energy is denoted by the symbol G U. This equation can be used to express the molar lattice energy of an ionic crystal in terms of molar lattice enthalpy, pressure, and change in volume in terms of the following variables: Difference between Lattice energy and Lattice Enthalpy 786 kilojoules is the amount of energy that must be supplied to one mole of sodium chloride in order for it to be separated into gaseous Na + and Cl – ions in this example.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
